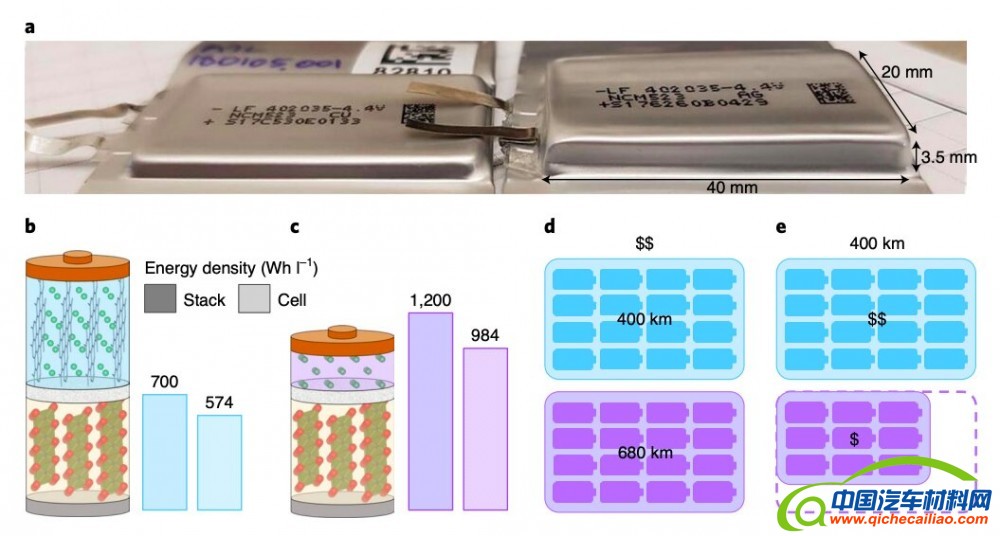
In addition to powering flying vehicles, high-energy cells (i.e., single units within a battery that convert chemical into electrical energy) could increase the distance that electric cars can travel before they need to be charged again. They may also reduce overall fabrication costs for electric vehicles, as similar results could be achieved using fewer but better-performing cells.
Anode-free lithium metal cells are particularly promising for creating batteries with higher energy densities. While they use the same cathode as Li-ion cells, these cells store energy via an electroplated lithium metal instead of a graphite host, and they can have energy densities that are 60% greater than those of Li-ion cells.
Despite their high energy densities, anode-free lithium metal cells typically have a lower capacity and short life cycles. This is primarily due to the fact that they derive all their lithium from the positive electrode and use no excess lithium. In addition, their safety has not yet been confirmed in experimental settings.
Researchers at Dalhousie University in Canada have been exploring ways in which the limitations of anode-free lithium metal cells could be circumvented or overcome. In a recent paper published in Nature Energy, they investigated the failure of anode-free cells they developed and proposed a possible solution that could prolong their' life cycle. Their paper is based on one of their previous works in which they explored the use of dual-salt carbonate electrolytes to extend the lifetime of anode-free lithium metal cells.
"Recently, we demonstrated long-lifetime, anode-free cells using a dual-salt carbonate electrolyte," the researchers wrote in their paper. "Here, we characterize the degradation of anode-free cells with this lean (2.6 g Ah-1) liquid electrolyte."
The researchers used two techniques, scanning electron microscopy and X-ray tomography, to identify the cause of failure in the anode-free cells they created. Their findings revealed that the morphology of the cells deteriorated over time.
They then investigated this deterioration further using two additional techniques: nuclear magnetic resonance spectroscopy and ultrasonic transmission mapping. Their findings suggested that the cause for the deterioration of the lithium inside the cells was the electrolyte, which became damaged and depleted over time.
In addition to investigating the reasons for the failure of the anode-free cells they created, the researchers carried out tests evaluating their safety. The observations they collected throughout their study allowed them to devise an electrolyte that optimizes the performance of their cells and extends their lifetime up to 200 cycles.
While the electrolyte developed by the researchers is a significant step forward in the development of high-energy batteries with longer lifetimes, for anode-free batteries to be implemented in real-world settings, these lifetimes would need to be extended further, as they are still not as good as those of more traditional lithium-ion batteries. In their next studies, the researchers will thus continue exploring ways in which they can significantly increase their cells' lifetime.